What Is the Opposite of Sublimation? (Simple Explanation + Printing Context)
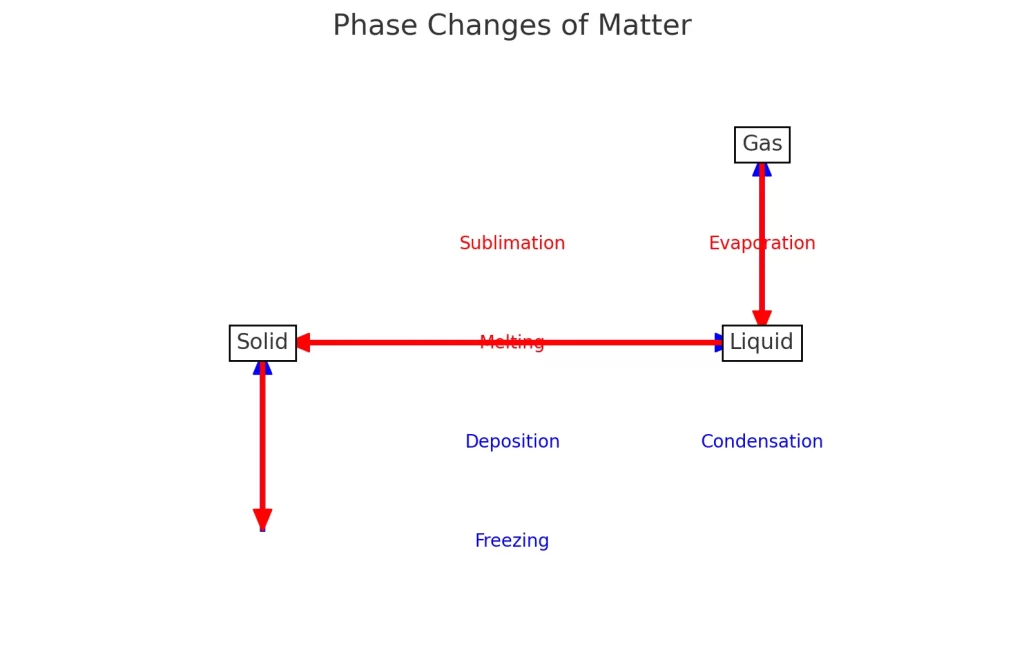
The opposite of sublimation is deposition, a process where a gas turns directly into a solid without becoming a liquid first.
But if you’re working with sublimation printing, understanding this concept isn’t just science. It helps you understand how heat, ink behavior, and material reactions actually work during the printing process.
In this guide, you’ll learn what deposition means, how it compares to sublimation, and why it matters when creating high-quality sublimation prints.
This is especially useful when troubleshooting print quality issues or working with different materials.
What Is the Opposite of Sublimation?
The opposite of sublimation is deposition, a process where a gas changes directly into a solid without becoming a liquid.
- Sublimation → Solid → Gas
- Deposition → Gas → Solid
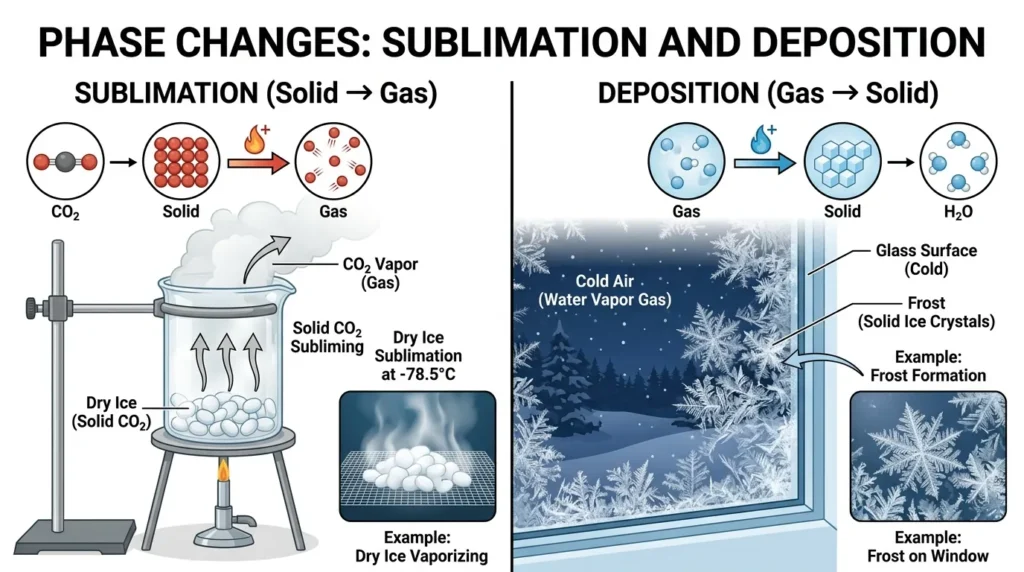
This direct phase change happens when energy is either added (sublimation) or removed (deposition).
What Is Deposition in Simple Terms?
Deposition happens when gas particles lose energy and form a solid.
Here’s a simple way to understand it:
- Gas cools down
- Molecules slow down
- They bond together into a solid
A common example is frost forming on a cold surface. Water vapor in the air turns directly into ice without becoming liquid water first.
Sublimation vs Deposition (Key Differences)
Here’s a clear side-by-side comparison:
| Feature | Sublimation | Deposition |
|---|---|---|
| Direction | Solid → Gas | Gas → Solid |
| Energy | Requires heat | Requires cooling |
| Phase Change | Skips liquid | Skips liquid |
| Example | Dry ice turning to vapor | Frost forming on glass |
If you want a deeper understanding of how sublimation works in real workflows, check this beginner sublimation printing guide.
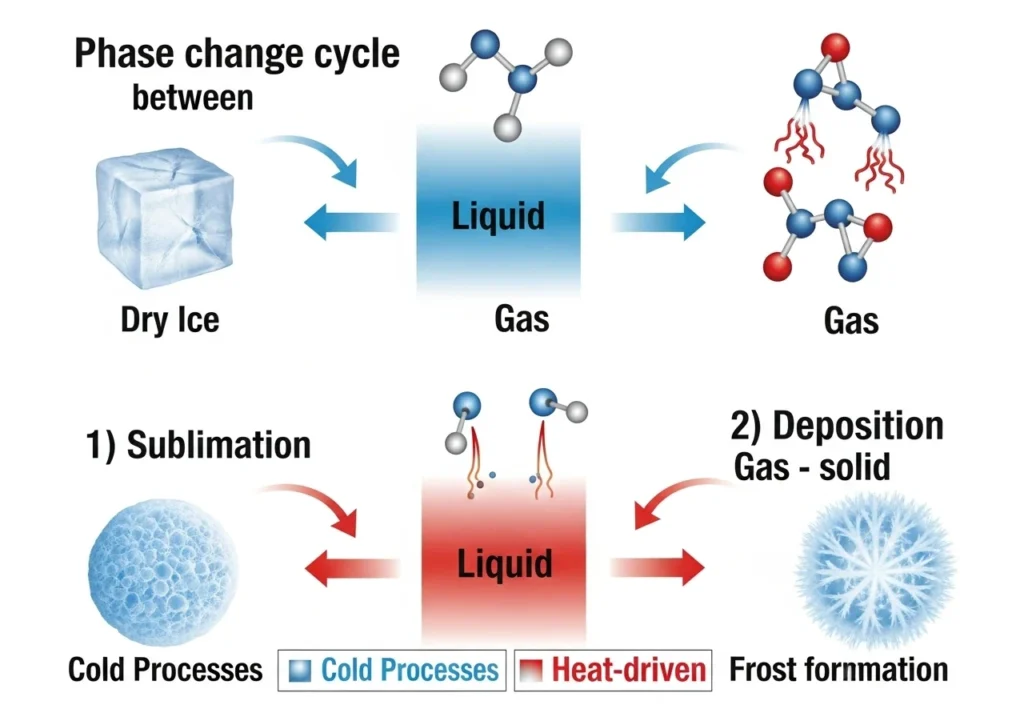
Real Examples of Deposition
Deposition happens more often than most people realize.
Frost Formation
Water vapor turns directly into ice on cold surfaces.
Snow Formation
Snowflakes form when water vapor deposits into ice crystals in clouds.
Freezer Frost
That icy buildup in your freezer is deposition over time.
Industrial Coatings
Deposition is used in manufacturing to create thin solid layers on surfaces.

How This Relates to Sublimation Printing
In sublimation printing, heat causes solid ink to convert into gas and bond with polyester or coated surfaces.
Deposition is the reverse process. While you don’t directly use deposition in printing, understanding it helps you:
- Understand temperature control
- Avoid poor ink transfer
- Recognize how gases behave under cooling
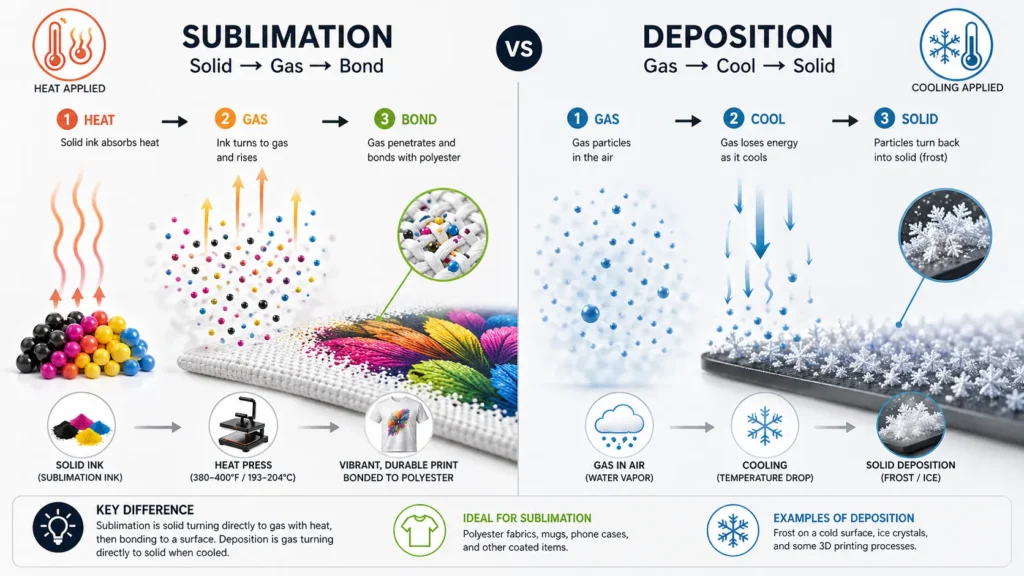
Why This Helps You Avoid Printing Mistakes
Understanding how sublimation and deposition work makes it easier to control:
Now that we clearly understand sublimation let’s explore its antithesis.
- Heat press temperature
- Pressing time
- Material compatibility
If your prints look faded or inconsistent, it often comes down to how well the sublimation process is controlled.
Why Sublimation and Deposition Matter in Printing
In sublimation printing, the process relies on solid ink turning into gas under heat, which then bonds with materials like polyester.
Understanding deposition helps you better understand the reverse behavior of gases and how temperature affects material transitions.
If you’re learning the full workflow, this complete sublimation process explained guide walks you through each stage step-by-step.
Common Confusion (Deposition vs Other Phase Changes)
Many beginners mix up deposition with other processes. Here’s how to avoid that:
Deposition vs Condensation
- Deposition: Gas → Solid
- Condensation: Gas → Liquid
Is Deposition Reverse Sublimation?
Yes, deposition is often called reverse sublimation because it is the exact opposite process.
Does Deposition Always Need Cold Temperatures?
Yes, deposition occurs when gas loses enough energy usually due to cooling.
To better understand key terminology like this, review these common sublimation printing terms explained.
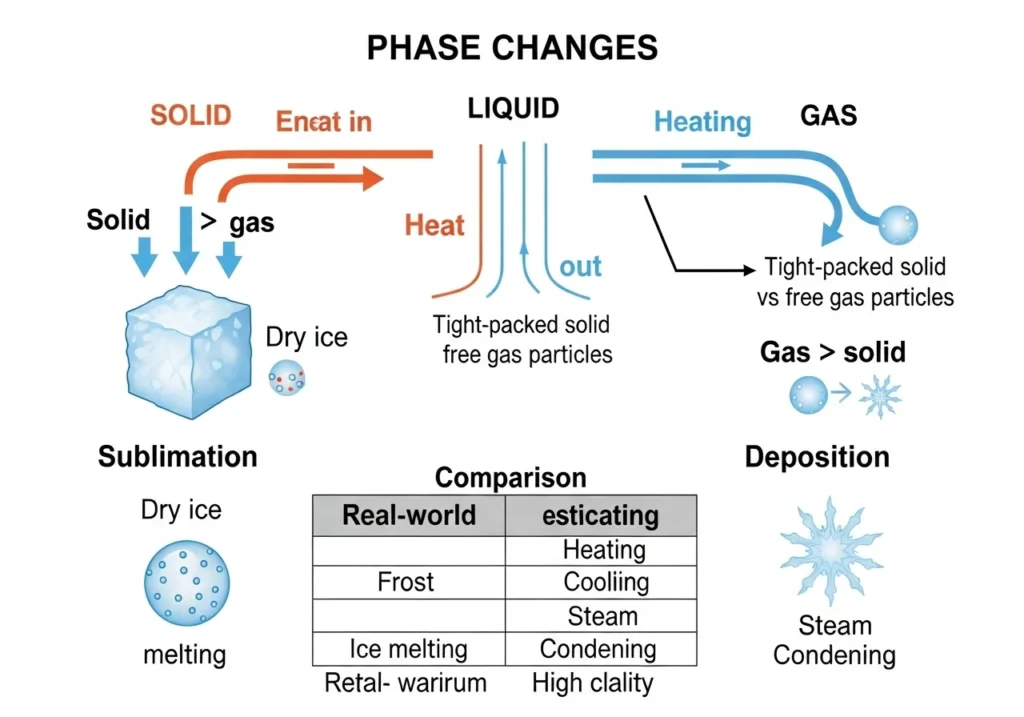
Simple Way to Remember
A quick way to keep this clear:
- Sublimation = heat added → solid becomes gas
- Deposition = heat removed → gas becomes solid
Think of it like this:
- Warm → particles spread out → gas
- Cold → particles come together → solid
Practical Tips to Understand This Better
If this still feels confusing, here’s a simple way to remember:
- Sublimation = heat added → solid disappears into gas
- Deposition = heat removed → gas becomes solid
Think of it like this:
- Warm → particles spread out → gas
- Cold → particles come together → solid
Frequently Asked Questions
What is the opposite of sublimation in chemistry?
In chemistry, the opposite of sublimation is deposition, where gas changes directly into a solid.
What is the opposite of sublimation in physics?
In physics, deposition is also the opposite of sublimation, as both describe energy-based phase changes.
What is the opposite of sublimation?
The opposite of sublimation is deposition, where a gas turns directly into a solid without becoming a liquid.
Is deposition the same as reverse sublimation?
Yes, deposition is often called reverse sublimation because it is the exact opposite process.
What is an example of deposition?
Frost forming on a cold window is the most common example of deposition.
Does deposition require heat?
No, deposition requires cooling. Gas particles lose energy and form a solid.
Is gas to solid sublimation?
No, gas to solid is deposition, not sublimation.
Quick Recap
Deposition is the opposite of sublimation, turning gas directly into a solid through cooling.
While sublimation drives the printing process, understanding deposition gives you a clearer picture of how temperature and material behavior affect your results.
Once you understand both, it becomes much easier to control your prints and avoid common mistakes with Subli Genius Print.








One Comment