Sublimation in Nature: Examples, Water Cycle & Simple Explanation
Sublimation in nature is something you’ve likely seen but never noticed. Snow disappears without melting, ice shrinks in your freezer, and frost forms overnight these are all signs of sublimation.
In simple terms, sublimation is when a solid turns directly into a gas without becoming liquid first. In this guide, you’ll learn how sublimation works in nature, how it fits into the water cycle, and real examples you can recognize in everyday life.
For a complete overview of how sublimation works beyond nature, explore this beginner sublimation printing guide.
What Is Sublimation in Nature?
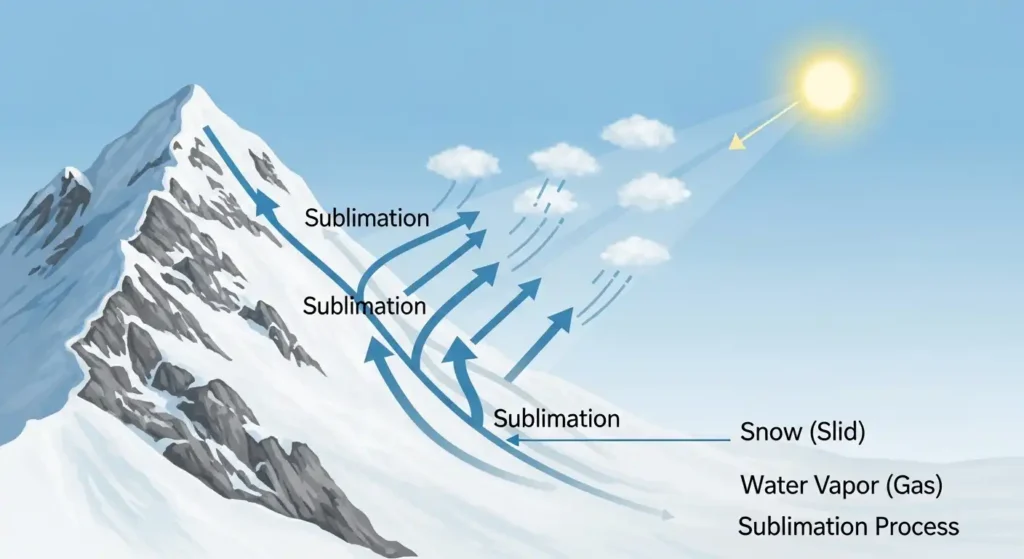
Sublimation in nature is the process where a solid like ice or snow changes directly into water vapor without passing through the liquid stage.
This usually happens in:
- Cold environments
- Dry air conditions
- Low-pressure areas (like mountains)
You may notice this on a cold, sunny day when snow slowly disappears without forming puddles.

How Sublimation Happens (Simple Explanation)
Sublimation occurs when environmental conditions allow solid particles to escape directly into the air as gas.
Key conditions include:
- Low humidity (dry air pulls moisture away)
- Cold temperatures (prevents melting)
- Sunlight or energy (provides enough heat for transition)
If these conditions are met, ice doesn’t melt it simply vanishes into vapor.
Sublimation in the Water Cycle
Sublimation plays an important role in the natural water cycle.
Quick answer:
Sublimation in the water cycle is when ice or snow turns directly into water vapor and enters the atmosphere without melting.
Where it commonly happens:
- Mountain regions
- Polar ice caps
- Glaciers
Instead of melting into liquid water, frozen water skips a step and becomes vapor. This vapor later contributes to cloud formation and precipitation.
For a deeper understanding of how phase changes work, you can explore the triple point and sublimation explained.

Real Examples of Sublimation in Nature
Here are clear, real-world examples you can easily understand:
1. Snow Disappearing on Mountains
Snow on high peaks can vanish without melting due to dry air and sunlight.
2. Glaciers Losing Ice
Glaciers shrink not only from melting but also from sublimation.
3. Polar Ice Caps
Even in freezing temperatures, ice can turn directly into vapor and enter the atmosphere.
4. Frost Formation (Reverse Process)
Water vapor turns directly into ice on cold surfaces this is called deposition.
5. Ice Cubes in Freezers
Ice slowly shrinks over time without turning into water.
You can explore more relatable cases in this guide on real-life examples of sublimation in daily life.
Step-by-Step: How Water Sublimates
Here’s a simple way to understand the process:
- Ice or snow is exposed to dry air
- Sunlight or heat provides energy
- Surface molecules gain enough energy to escape
- Solid turns directly into water vapor
- Vapor mixes into the atmosphere
This process happens slowly but continuously in the right conditions.
Why Sublimation Matters in Nature
Sublimation is not just a scientific concept it has real environmental impact.
It affects:
- Water cycle balance
- Glacier size and movement
- Climate patterns
- Atmospheric moisture levels
In many cold regions, sublimation is a major factor in how water moves through ecosystems.
Common Misunderstandings About Sublimation
❌ “Ice always melts before turning into vapor”
Not true. Sublimation skips the liquid stage completely.
❌ “Sublimation only happens in labs”
It happens naturally in mountains, glaciers, and even your freezer.
❌ “It requires extreme heat”
Actually, sublimation often happens in cold but dry conditions.ate to a gas, releasing a smell that keeps moths and other insects at bay.
Quick Facts About Sublimation
- It is a direct solid-to-gas phase change
- Common in cold, dry climates
- Plays a role in the water cycle
- Happens in both nature and everyday life
- The reverse process is called deposition
How to Observe Sublimation Yourself
You don’t need a lab to see sublimation.
Try this simple method:
- Place ice cubes in your freezer uncovered
- Leave them for several days
- Notice how they shrink without melting
That’s sublimation happening in your home.

Frequently Asked Questions
Can water really sublimate in nature?
Yes, ice and snow can turn directly into vapor in dry, cold environments.
Do I need heat for sublimation to happen?
You need some energy, but it doesn’t require high temperatures sunlight is often enough.
Is sublimation part of the water cycle?
Yes, it allows frozen water to enter the atmosphere directly as vapor.
What’s the difference between sublimation and evaporation?
Sublimation skips the liquid stage, while evaporation happens from liquid to gas.






One Comment